Limitations

Boron is not present in nature in its elemental form.
It can only be found in nature as a compound; the most abundant being sodium tetraborate decahydrate (borax) which ores in few countries around the world.
It can only be found in nature as a compound; the most abundant being sodium tetraborate decahydrate (borax) which ores in few countries around the world.
For the definition of ore:
Health

Boron is very beneficial to a humans; proving them with necessary nutrients. However large quantities of this element can cause problems such as
<---poisoning.
When an organism consumes an extensive amount of Boron the organism can encounter various problems.
<---poisoning.
When an organism consumes an extensive amount of Boron the organism can encounter various problems.
- The human body can handle no more than 1-10 mg of boron each day. If the daily limit consumed is exceeded, it can eventually lead to problems, such as dis-function of the reproductive organs.
- The intake of too much boron can also affect the stomach, liver, kidneys, brain, and can potentially cause death.
- If a pregnant women intakes an unreasonable amount of Boron, her baby is very likely to suffer birth defects and/or delayed development.
-Boron is found concentrated in the spleen, bone, and thyroid (the thyroid being part of the Endocrine System with the responsibility to produce hormones). Thus implying Boron’s functions in bone metabolism and suggesting a potential role for Boron in hormone metabolism.
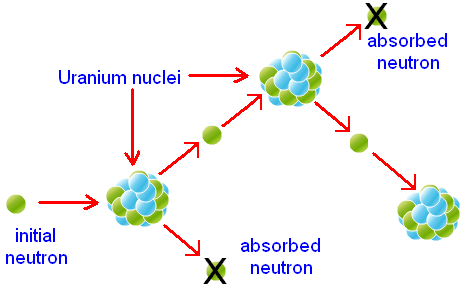
Controlling Nuclear Reaction Limitations

Although Boron is used as a nuclear reaction stabilitator, keeping the fission of Uranium (U-235) under control, it does have mechanical properties that are less than desirable for building a control rod structure. This is because Boron is a brittle, salt-like material.
Because of its weak properties, Boron may need to be enriched to reach the necessary absorptivity, in order to do its job.
Some methods for working around this situation, are to use a steel alloy enriched with Boron- such as the one depicted above. Although the most commonly used alloy in nuclear fission is one with Silver (Ag) and Indium (In), giving good mechanical properties and a more uniform absorption spectrum.
Because of its weak properties, Boron may need to be enriched to reach the necessary absorptivity, in order to do its job.
Some methods for working around this situation, are to use a steel alloy enriched with Boron- such as the one depicted above. Although the most commonly used alloy in nuclear fission is one with Silver (Ag) and Indium (In), giving good mechanical properties and a more uniform absorption spectrum.