Lily Heller
Period 2
12-6-13
Period 2
12-6-13
BORON

Atomic Number: 5
Atomic Mass: 10.81 amu
Density : 2.34 g/cm-3
Melting Point: 2076 degrees Celsius
Boiling Point: 3927 degrees Celsius
Atomic Radius: 0.098 nm
Ionic Radius: 0.027 nm
Number of Isotopes: 2 (Boron -11 and Boron -10)
Oxidation State: +3
Electron Configuration: [He] 2s2 2p1
Discovered By: Sir Humphry Davy and J.L. Gay- Lussac
Atomic Mass: 10.81 amu
Density : 2.34 g/cm-3
Melting Point: 2076 degrees Celsius
Boiling Point: 3927 degrees Celsius
Atomic Radius: 0.098 nm
Ionic Radius: 0.027 nm
Number of Isotopes: 2 (Boron -11 and Boron -10)
Oxidation State: +3
Electron Configuration: [He] 2s2 2p1
Discovered By: Sir Humphry Davy and J.L. Gay- Lussac
General facts
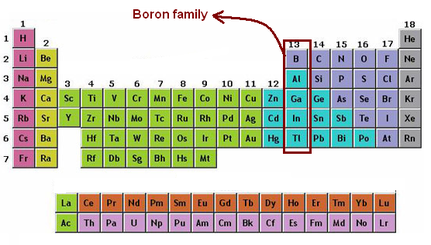
Is part of the Boron Family and is the only metalliod in this group. Being a metalliod means that Boron has properties that both metals and non metals have.
-At standard temperature Boron is a poor conductor
-At high temperatures Boron is a good conductor
-At standard temperature Boron is a poor conductor
-At high temperatures Boron is a good conductor
Boron occurs in the environment through the natural process of weathering. However the releasing of this element done by humans is the reason why it is found in the environment. Humans add Boron to the environment because it does not occur naturally in its elemental form.
Boron is released into the air, soil, and water. Once this element is released and weathered into the various form of matter, it is then absorbed and is affected in many different ways.
Boron is released into the air, soil, and water. Once this element is released and weathered into the various form of matter, it is then absorbed and is affected in many different ways.

Humans are exposed to Boron through food, such as fruit (especially grapes) and green vegetables, grains, water, air, and consumer products.

Boron is very similar to Carbon because of its capability of forming stable covalent bonds .
